First Law of Thermodynamics It states that the heat and work are mutually convertible. Third law of thermodynamics.

Solved Match Each Law Of Thermodynamics To Its Appropriat Chegg Com
In the next few sections we will discuss each of the laws of thermodynamics in detail.
Thermodynamics and its laws. 2nd Law of Thermodynamics The Second Law of Thermodynamics states that the state of entropy of the entire universe as an isolated system will always increase over time. Second law of thermodynamics is used to check whether the process is spontaneous or not. Types Of Thermodynamics laws And Its Application 1.
The First Law of Thermodynamics The first law of thermodynamics thinks big. The Laws of Thermodynamics Entropy and Gibbs Free Energy - YouTube. The laws of thermodynamics govern the direction of a spontaneous process ensuring that if a sufficiently large number of individual interactions like atoms colliding are involved then the direction will always be in the direction of increased entropy.
Various sources show the following three potential formulations of. The laws of thermodynamics are important unifying principles of biology. The third law of thermodynamics is essentially a statement about the ability to create an absolute temperature scale for which absolute zero is the point at which the internal energy of a solid is precisely 0.
The state of the entropy of the entire universe as an isolated system will always. Second law of thermodynamics. 3rd Law of Thermodynamics.
Thermodynamics is the branch of physics which deals with the study of relation bw heattemperaturework and energy. When two systems are each in thermal equilibrium with a third system the first two systems are in thermal equilibrium with each other. Ie energy can neither be created nor destroyed but it can convert into another form of energy.
Second law of thermodynamics. The Second Law of Thermodynamics. How many laws of thermodynamics are there.
The most important laws of thermodynamics are. Entropy of a. Put another way the First Law of Thermodynamics states that energy cannot be created or destroyed.
First law of thermodynamics. First law of thermodynamics-. The zeroth law of thermodynamics.
Laws of Thermodynamics First law of thermodynamics. The second law also states that the changes in the entropy in the universe can never be negative. The FLT defines internal energy as a state function and provides a formal statement of the conservation of energy.
Second law of thermodynamics. It is the law of conservation of energy. This property makes it meaningful to use thermometers as the third system and to define a temperature scale.
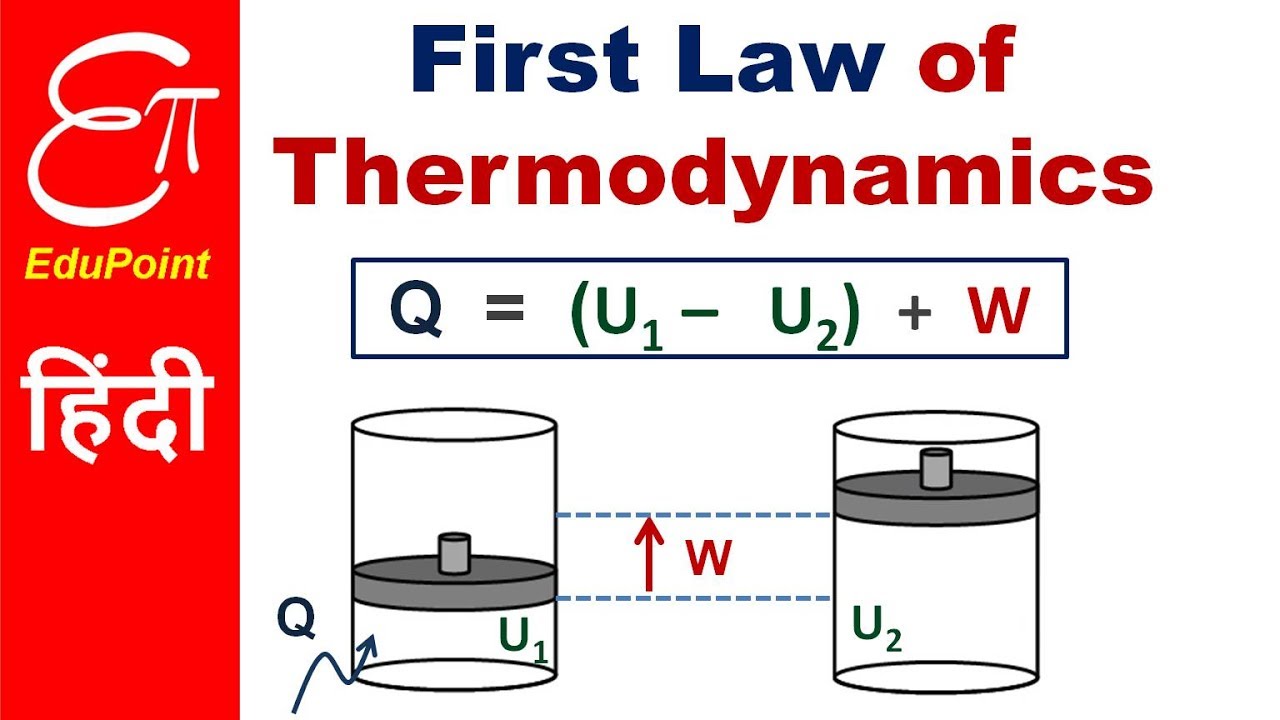
The first law of thermodynamics is the law of the conservation of energy which states that although energy can change form it can be neither be created nor destroyed. First Law of Thermodynamics. The Year Earth Changed Apple TV.
When energy moves into or out of a system the systems internal energy changes in. Zeroth law of thermodynamics states that when two systems are each in thermal. Whether the process occurs on its own or not It also tells us the direction in which the process will proceed.
Third law of thermodynamics. These principles govern the chemical processes metabolism in all biological organisms. First law of thermodynamics concerns principle of conservation of energy.
Why do we use the 2nd law of thermodynamics. The First Law of Thermodynamics also known as the law of conservation of energy states that energy can neither be created nor destroyed. There are 3 laws of Thermodynamics.
If playback doesnt begin. Zeroth law of thermodynamics. Zeroth law of thermodynamics-.
It deals with the total amount of energy in the universe and in particular it states that this total amount does not change.
What Are The First 2 Laws Of Thermodynamics How Are They Used Quora
